|
|
|

|
O-003 / O-003-1ML updated item number format
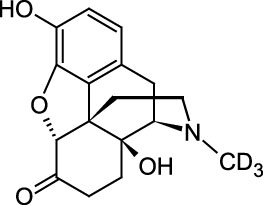
Oxymorphone-D3
100 ug/mL in Methanol | Certified Reference Material
|
|
A stable-labeled internal standard suitable for quantitation of oxymorphone levels in urine, serum, or plasma by LC/MS or GC/MS for urine drug testing, pain prescription monitoring, clinical toxicology, forensic analysis or pharmaceutical research. Oxymorphone, sold under trade names such as Opana, Numorphan, and Numorphone, is an opiate analgesic indicated for the relief of moderate to severe pain.
|
|
|
|
|
| Concentration:
|
100 ug/mL
|
| Solvent:
|
Methanol
|
| Unit Size:
|
1 mL/ampoule
|
| CAS Number:
|
145225-03-2
|
| Chemical Formula:
|
C17H16D3NO4
|
| Molecular Weight:
|
304.36
|
| Certificate of Analysis:
|
|
| US List Price:
|
|
|
|
Regulatory Control
▶ USDEA exempt chemical preparation - no USDEA registration or paperwork required
▶ Canada Test Kit Registration # 61-1415 - no Health Canada import authorization required
read more about ordering regulated substances
UN Number, Class, PG
1230, 3, II
Safety Data Sheet
HS Code
2845.90.0100
|
▶ A High-throughput Pain Management Panel in Urine Using IONICS 3Q 120 LC-MS/MS and an SLE Sample Prep Platform | IONICS Mass Spectrometry Group inc., 2014
|
|
▶ Automated Hydrolysis, DPX Extraction and LC/MS/MS Analysis of Pain Management Drugs from Urine | Fred D.Foster, John R. Stuff, Edward A.Pfannkoch, GERSTEL, 2014
|
|
▶ Development of a High-Throughput LC-MS/MS Assay for a Pain Management Panel from Urine | IONICS Mass Spectrometry Group inc., 2014
|
|
▶ Development of a high-throughput LC-MS/MS assay for pain management panel from urine | Hui Qiao, Changtong Hao, Sha Joshua Ye, Lisa Cousins, IONICS Mass Spectrometry Group inc., 2014
|
|
▶ High Throughput Screening and confirmation of 41 Pain Panel Drugs in Oral Fluid by an Integrated On-Line Extraction UHPLC-MS/MS System | Louis Maljers, Zicheng Yang, Bruker Daltonics Inc, 2016
|
|
▶ LC/MS (TOF) Analysis of Opioid Drugs and Glucuronide Metabolites in Urine on Ascentis® Express 2.7 µm HILIC after SPE using Supel™-Select HLB, ß-Glucuronidase Enzyme Digestion | Sigma-Aldrich®, 2014
|
|
▶ LC/MS Analysis of Opioid Glucuronide Metabolites in Urine on Ascentis® Express 2.7 µm F5 after Solid Phase Extraction (SPE) using Supel™-Select HLB | Sigma-Aldrich®, 2014
|
|
▶ Proof of Concept for Automated SPE/HPLC/MS/MS Methods to Replace Traditional Immunoassay with MS Confirmation of Driving Under the Influence Samples | Robert M. Sears, Toxicology Technical Leader , Kenneth Lewis, CEO , and Kim Gamble, President, ITSP Solutions, INC., 2014
|
|
▶ Proof of Concept/Comparison of Automated SPE/HPLC/MS/MS Methods to Traditional Immunoassay with MS Confirmation in Death Investigation Cases | Robert M. Sears, Wendy C. Bell , Kenneth C. Lewis, Thurman L. Allsup and Kim Gamble, ITSP Solutions, INC., 2014
|
|
|
|
|